A decade of research is paying off as first patient on the East Coast is now undergoing innovative rehabilitation after having a brain stimulation device implanted by surgical team at Mass General Hospital
Every hour, nearly 100 Americans experience a stroke and for most, a long and difficult road to partial recovery will follow. Occupational, physical, and speech-language therapy can help, but a patient’s rehabilitative progress often reaches a plateau.
Now, there’s new hope, and Teresa Kimberley, PhD., researcher and director of the Brain Recovery Lab at the MGH of Health Professions, is playing a key role.
An FDA-approved, drug free approach – a brain stimulation device connected to the vagus nerve, paired with rehabilitation therapy – may improve hand and arm function by 200 – 300% above previous levels. The pairing of therapy with vagus nerve stimulation is helping the brain create or strengthen new neural pathways - like building a bridge to bypass a damaged area.
This breakthrough in stroke therapy is happening thanks to the collaborative efforts of Mass General Brigham’s research and clinical institutions.
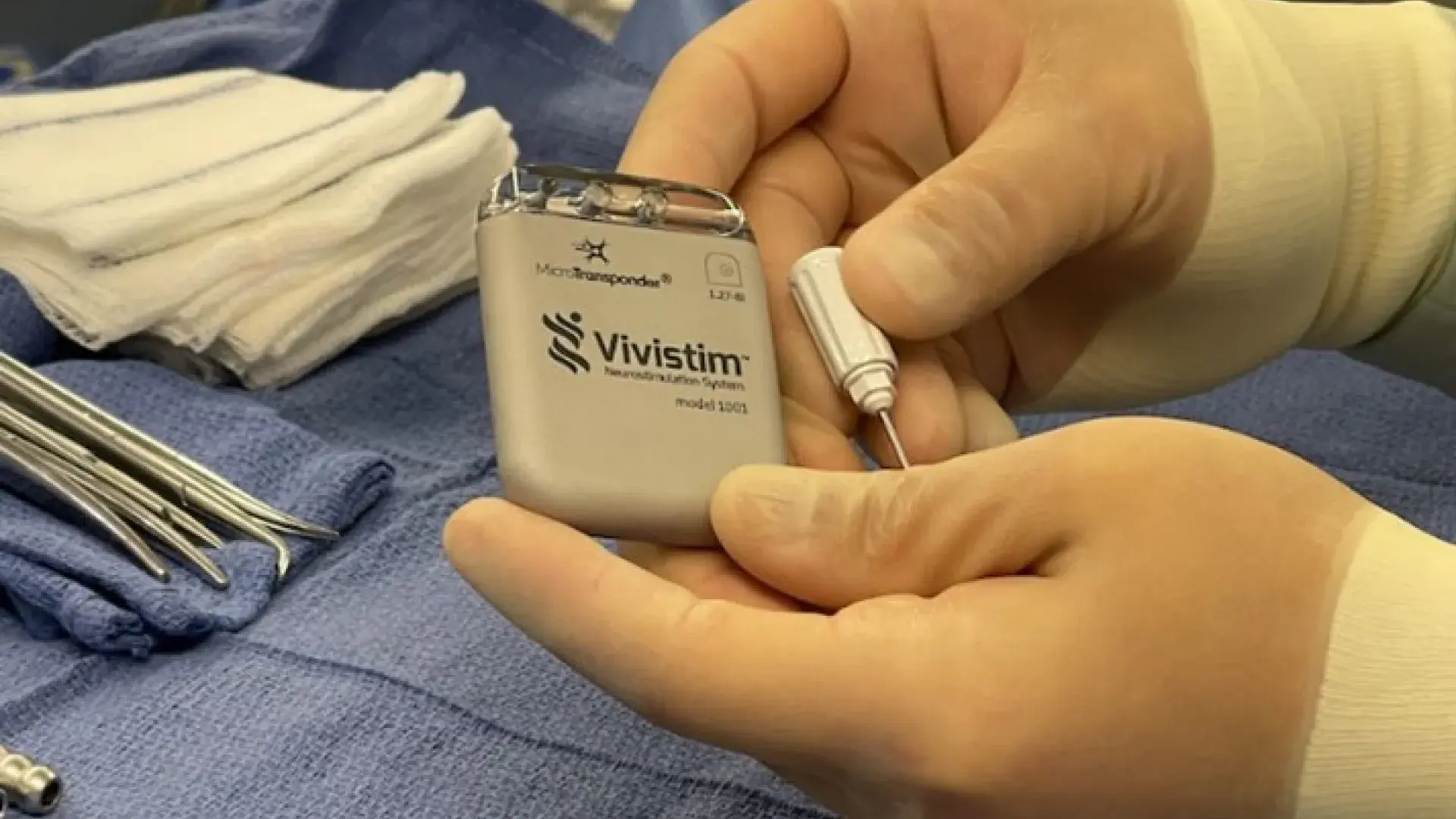
Dr. Kimberley developed the rehabilitation protocol the FDA says is necessary to pair with the groundbreaking technology called the Vivistim® Paired VNS™ System, which employs vagus nerve stimulation (VNS) during rehabilitation therapy to improve upper extremity motor function. Research shows that a stroke survivor will only see significant progress if the rehabilitation and technology are used together.
“This is a watershed moment for rehabilitation science,” said Dr. Kimberley. “This is the first time that brain stimulation combined with rehabilitation therapy for stroke is available for patients beyond a research study. There is a new FDA-approved option that has strong potential to improve hand function. It is very exciting.”
The first step for this new therapy took place in late August when the first patient on the East Coast had the small Vivistim device placed around a nerve in his neck (the vagus nerve) and under the skin of his upper left chest area during an outpatient procedure at Massachusetts General Hospital. The procedure was performed by Ziv Williams, MD, a neurosurgeon at Mass General and Faculty in the Program in Neuroscience at Harvard Medical School, under care being directed by David Lin, MD, a neurologist, Faculty in the Center for Neurotechnology and Neurorecovery, and Director of the Mass General Neurorecovery Clinic.
The patient, 40-year-old David Sullivan of Concord, MA, suffered a stroke in 2020, leaving him with aphasia and limited use of his left arm, hand, leg, and foot. Two years of physical therapy, occupational therapy and speech therapy hadn’t enabled him to do the daily activities he desired, so he jumped at the opportunity to begin Paired VNS Therapy.
“It’s definitely exciting to be involved with this kind of cutting-edge technology,” said Sullivan, a father of two young girls. “I’ve tried everything these past two years and have left no stone unturned. The fact this is so new makes me a little anxious, but I know I’m in good hands. Knowing the device and intense rehabilitation give me a good chance to regain more hand and arm movement is a wonderful thing.”
“This is an exciting development in stroke rehabilitation,” said Lin. “The fact that this new treatment pairs device-based neural stimulation with rehabilitation provides a new paradigm for neurologically based therapies in stroke recovery. As a clinician, I find it very rewarding to see a therapy that has been based on decades of fundamental neuroscience now be translated to help patients with stroke.”
Sullivan is currently undergoing outpatient rehabilitation at Spaulding Rehabilitation Hospital.
Mass General is a certified Advanced Comprehensive Stroke Center, capable of treating the most complex stroke cases. “I am excited about this development and hope we can offer more of the novel and effective recovery interventions to our stroke patients,” said Natalia Rost, MD, Mass General Stroke Service Chief.
When used in conjunction with in-clinic rehabilitation therapy, the Vivistim System can help increase neuroplasticity for survivors of ischemic stroke so that their brain relearns how to perform daily activities that were routine prior to their stroke.
Vivistim is manufactured by MicroTransponder®, a global medical device company.
How it Works
While VNS has been an extensively researched treatment option for epilepsy and depression, this is its first application in rehabilitation therapy.
“If you pair a pulse of the vagus nerve stimulation with each repetition of intense physical or occupational therapy, the outcome is enhanced,” says Kimberley. “With each repetitive movement there is likely a meaningful effect in the brain that ultimately helped people in our study improve twice as much as they did with the therapy alone.”
“Arm motor recovery after stroke is often incomplete. This new modality is designed to help improve patients’ recovery of functional use of the affected arm after stroke,“ said Randie Black-Schaffer, MD, MA, Medical Director of the Stroke Program at Spaulding Rehabilitation Hospital and Chief of the Division of Stroke and Neurology of the Dept. of Physical Medicine and Rehabilitation at Harvard Medical School.
“When you stimulate the Vagus Nerve, the brain is firing more than it would on its own,” said Dr. Kimberley. “The VNS enhances neurotransmitter release to help create connections, or pathways, that are critical in getting the brain to perform a specific function, like picking up that pen. The VNS appears to give the brain a way to lay down these pathways permanently. If patients keep using these pathways, while reinforcing with therapy and home practice, these abilities will remain and get stronger.
The promise of increased functionality is based on a two-year clinical trial Kimberley and others on the Mass General Center for Neurotechnology and Neurorecovery (CNTR) team recently finished, the results of which were published in The Lancet. “The translation of novel research, from concept through clinical trials to clinical practice, is central to our mission at CNTR,” said Lin.
The clinical studies have shown that after in-clinic therapy, the Vivistim Therapy increases hand and arm function by two to three times, as compared to rehabilitation therapy alone.
That improved movement is what Sullivan is banking on. Since losing partial functionality on his left side, the lefthander hasn’t been able to write, throw a ball with his children, ride a bike, golf, or play his guitar. He desperately wants those abilities back, along with the chance to go back to work as a software sales operations director.
“I’ve got some interests that motivate me, for sure,” said Sullivan. “I’m already doing OT, PT, and speech therapy anyway, but the promise of this new approach – the device combined with the rehabilitation – gives me hope.”
Therapy Training is Critical for Success
While the VNS device is on the market to be sold to any hospital that wants it, the effectiveness will be muted unless it’s combined with the therapy protocol outlined by Dr. Kimberley.
“It’s not as simple as implanting this device. Medical centers will need to follow the protocol that we've outlined, and teams will need to be equipped with the right therapy training to challenge these patients for the best outcome possible,” said Kimberly. “We need the people who are trained to do that properly.”
Dr. Kimberly developed the rehabilitation protocol that the VNS is enhancing. She created a rigorous process that puts patients on the cusp of, “I can still do it” without so frustrated that they want to quit. She says the key is to find activities that will drive the brain’s neural patterns.
“If you're doing something that's right on the edge of your ability, it's going to make that better. If I'm a runner, and I only run three miles every day around the same loop, I'm never going to get better at running - I'm just going to maintain my running ability. But if I want to get better, I must do intervals or I must go longer or I must do something different. So, you have to do it where it's hard.”
For Dr. Kimberley, years of effort have finally paid off: she researched VNS for a decade, had a long-standing scientific consultation relationship with the company developing the technology, brought the trial to MGH, was the principal investigator running the trial, built the team to implement it, was senior author on the Lancet paper that published the results, and now is working to bring this as a clinical offering to MGB patients.
“I can’t tell you how many hours I have put into this through the years,” said Kimberley. “To see the progress we’ve made, to see a patient who will soon enjoy the benefits of the technology and rehabilitation, makes it all worth it. This is going to change the lives of so many patients, and that’s what research is all about, isn’t it?”
Do you have a story the Office of Strategic Communications should know about? If so, email ihposc [at] mghihp.edu.